Last Updated: 30 June 2022

Many fish and shrimp species are found easily infected by the white spot syndrome virus (WSSV). Death of these infected species causes such considerable loss to both ecosystem and Thailand’s economy at present. The need to study and understand molecular mechanism of WSSV infection is very important to development of an antiviral drug and control spread of the disease.
The previous study showed that VP37 envelope protein is generated by the virus DNA and essential for virus living. The VP37 engulfs the virus DNA and attaches to host cell to replicate the virus inside. At the end of VP37 amino acid sequence is the C-terminal domain responsible for binding WSSV to molecules of shrimp blood cell and heparin (in which heparin prevents blood clots). This binding cannot occur when gluing a group of sulfate molecules with negative charge such as sulfate galactan to C-terminal domain instead. It will be impossible for VP37 to attach to heparin and for the virus to replicate itself.

WSSV-infected fish with white spots as indicated by the red arrow
In this study, researchers created 3D-structure picture of C-terminal domain in VP37 by applying Synchrotron Protein Crystallography technique. The picture showed position views of two amino acids including Arginine at position 213 (R213) and Lysine at position 257 (K257) highly suspected as important binding sites to sulfate molecules in host cells. These two amino acids have positive charges and can attach to negative-charged heparin molecules for virus replication in hosts.
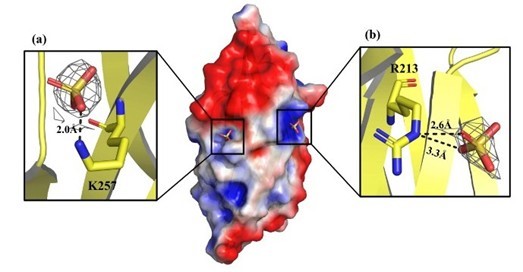
A 3D-structure picture of C-terminal domain in VP37 enveloped protein of WSSV
In this study, both R213 and K257 amino acids were modified to have neutral charge to study binding reaction of modified VP37 to heparin molecules. The researchers noticed no binding of the modified R213 and K257 to heparin, confirming the importance of the two amino acids as binding sites to sulfate molecules of heparin. The research result will be beneficial to development of antiviral drug in the future.
Article by:
Dr. Jakrada Attarataya, Beamline Scientist
Source:
Somsoros, W., Sangawa, T., Takebe, K., Attarataya, J., Wongprasert, K., Senapin, S., Rattanarojpong, T., Suzuki, M. & Khunrae, P. (2021). Crystal structure of the C-terminal domain of envelope protein VP37 from white spot syndrome virus reveals sulphate binding sites responsible for heparin binding. Journal of General Virology, 102(6). https://doi.org/10.1099/jgv.0.001611

